Extended X-Ray Absorption Fine Structure
Description
(This description of EXAFS was copied off the National Synchrotron Light Source website. You can find a link to the page on the navigation bar above.)
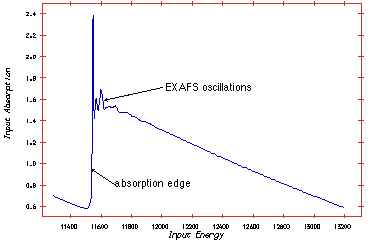
A monochromatic x-ray beam is directed at the sample. The photon energy of the x-rays is gradually increased such that it crosses one of the absorption edges of the elements contained within the sample. Below the absorption edge, the photons cannot excite the electrons of the relevant atomic level, but when the photon energy is just sufficient to excite the electrons, then a large increase in absorption occurs. The excited electrons have a low kinetic energy and can be backscattered by the atoms surrounding the emitting atom. The backscattering of the photoelectron affects slightly whether the X-ray photon is absorbed, so the probability of x-ray absorption will depend on the photon energy. The net result is a series of oscillations that can be used to determine the atomic number, distance and coordination number of the atoms surrounding the element being examined.